Collectively, our central interest is in the comprehensive mapping of the immune response to inflammation and infectious disease at the single-cell level. We seek to apply multiple high-dimensional cytometry, single-cell, analysis approaches to facilitate a ‘systems immunology’ approach to our investigation. A particular focus of our group has been the study of immunopathology, where the immune response to infection may be a significant driver of disease.
Featured research areas:
SARS-CoV-2 and COVID-19
|
SARS-CoV-2 and COVID-19 research The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic has caused >101 million infections and 2.18 million deaths worldwide (as of January 28, 2021). Infection with SARS-CoV-2 results in a spectrum of clinical presentations, called coronavirus disease 2019 (COVID-19), ranging from asymptomatic to fatal disease. Our team began investigating the immune response to COVID-19 in March 2020, in collaboration with researchers at the Doherty Institute and the University of Melbourne.
|
|
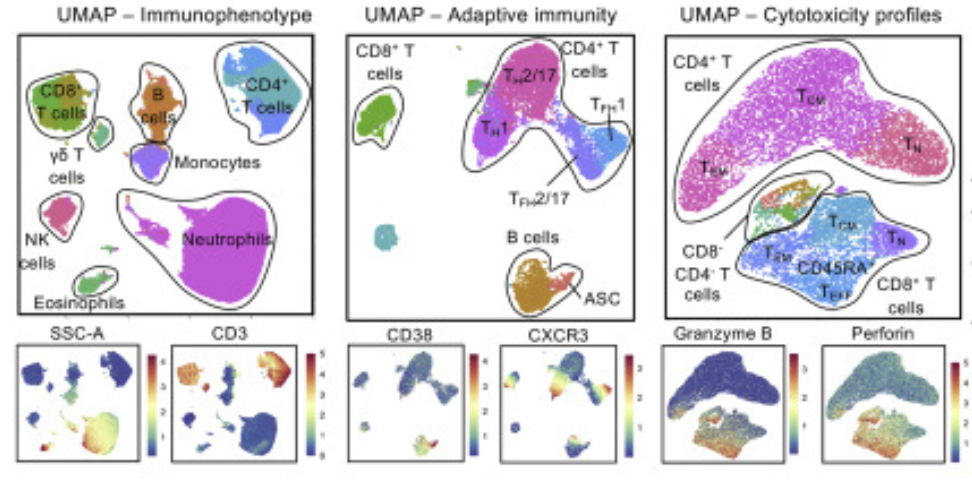
Mapping the immune dynamics of COVID-19 Our team played a key role in one of Australia's early COVID-19 immune profiling studies, led by the Kedzierska laboratory at the Doherty Institute (Koutsakos et al. Pre-print: Oct 2020, published in Cell Reports Medicine: Feb 2021). We developed and applied high-dimensional analysis tools to help comprehensively map the immune response to COVID-19, including a novel time-series analysis approach called TrackSOM. This study demonstrated that patients with severe disease exhibited an excessive and hyper-activated immune response, and was the first to show that T follicular helper cell responses could predict subsequent antibody levels, including neutralising activity of the antibody, a critical feature important for virus clearance and protection from infection.
|
|
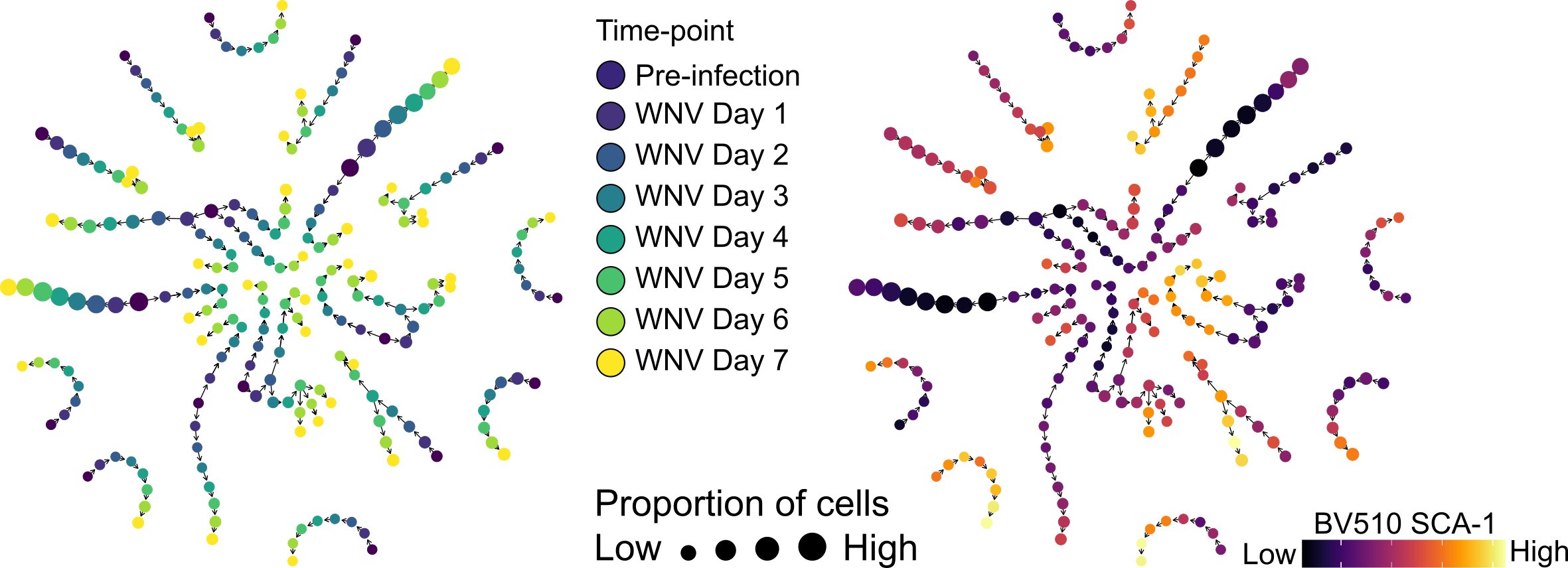
Ongoing COVID-19 research Our ongoing work in COVID-19 is interested in developing a more comprehensive understanding of how the immune response to SARS-CoV-2 leads to protection or immunopathology, with a particular focus on the role of the myeloid and innate immune system. Seminar, Leiden University Medical Center |
Viral encephalitis
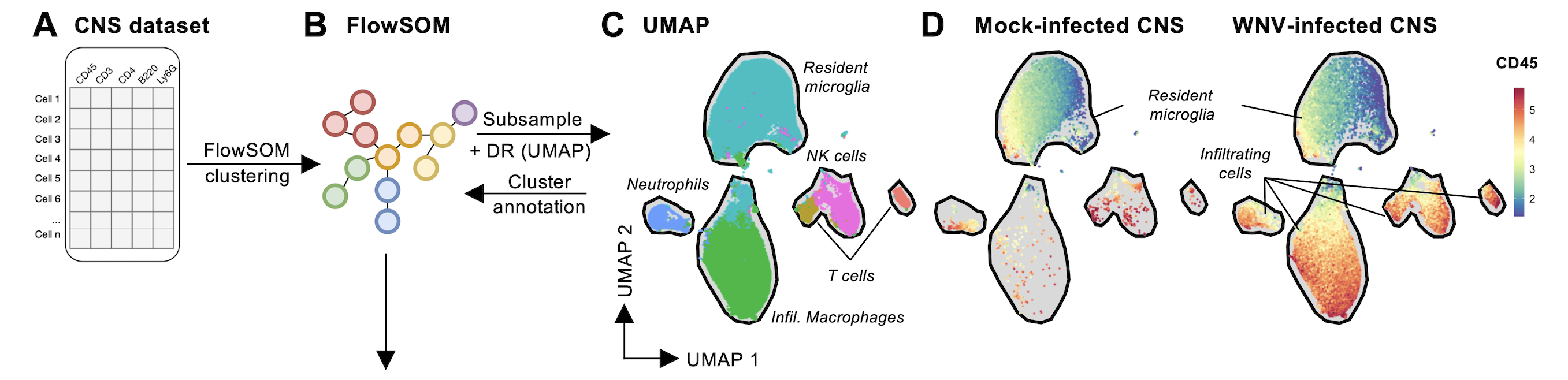
In mouse models of viral encephalitis, inflammatory monocyte-derived macrophages which infiltrate into the brain contribute significantly to disease (Getts et al. 2012), where immune-modulatory therapies targeting inflammatory monocytes may inhibit this process (Getts et al. 2014). Our recent work has comprehensively profiled the immune infiltrate in models of viral encephalitis, revealing complex cellular kinetics in both Zika virus (ZIKV) (Hayashida et al. 2019) and West Nile virus (WNV) (Ashhurst et al. 2020) encephalitis. Additionally, we have demonstrated that viral encephalitis drives an inflammatory mobilisation of the haematopoietic system in the bone marrow, resulting in the generation of pathogenic monocytes (Ashhurst et al 2019, Ashhurst 2020, Fluidigm webinar).
Viral and general immunology

Through various collaborations, we have applied our approaches in a number of areas, including T cell exhaustion in LCMV infection (Huber et al. 2017), VZV infection of NK cells (Campbell et al. 2018), antibody-mediated cell subset depletion (Jung et al. 2018), interferon-mediated lethality in LCMV infection (Jung et al. 2020), B cell responses in multiple sclerosis (Marsh-Wakefield et al. 2020), and intrapulmonary vaccination strategies (Ferrell et al. 2021).
